

executive clinical governance
CLINICAL PROGRAMME LEADERSHIP
Senior-level ownership and delivery accountability
for US-headquartered and European biotech sponsors expanding into Europe and adjacent markets —
from feasibility through CSR.
I provide accountable European
programme ownership across Phase
I–IV, ensuring governance clarity, execution control and predictable delivery — without building permanent European infrastructure.
programme ownership across Phase
I–IV, ensuring governance clarity, execution control and predictable delivery — without building permanent European infrastructure.
WHEN EUROPE IS RUNNING — BUT NO ONE OWNS IT


Senior EU leadership without infrastructure
Biotech sponsors expanding across the wider European region often encounter a governance misalignment: infrastructure exists, activity is visible — but executive-level ownership aligned with headquarters expectations is absent.
investor pressure
without delivery transparency
escalation-based management
instead of governance
hidden risks
during critical phases
fragmental decision
across countries and vendors
cro activity
but no true sponsor control
"European programmes rarely fail due to insufficient activity. They fail when executive accountability is unclear.
I am engaged when governance, risk visibility, and investor confidence must be protected."
I am engaged when governance, risk visibility, and investor confidence must be protected."
services
outsourced clinical talent
when engaged
- Immediate regional execution capacity
- Vacant or delayed leadership hires
- Programme timelines incompatible with traditional recruitment cycles
what i provide
- Access to pre-vetted European-region clinical professionals
- Rapid mobilisation
- Flexible FTE or fractional engagement models
- Single contractual and payment interface
A structured access model —
not a recruitment agency, and not a CRO replacement.
independent board & investor advisory
when engaged
- Pre-financing or partnering decisions
- Independent validation of timelines and feasibility
- CRO performance uncertainty
- Board-level need for unbiased risk clarity
what i provide
- Independent execution risk assessment
- Validation of timeline, cost, and country strategy
- Identification of hidden exposure before capital events
- Decision-ready insight for go / no-go clarity
"Structurally independent. No vendor revenue linkage. No operational bias."
Interim / Contract European-region Programme Director
when engaged
- European-region programme active but ownership unclear
- CRO underperformance or misalignment
- Value-critical Phase II/III or submission preparation
- Interim leadership gap
- Audit or inspection exposure
- Rapid scale-up or transition phase
what i provide
- End-to-end accountable European-region Programme Ownership
- Single decision and escalation authority
- Governance anchor across countries and vendors
- CRO oversight with performance enforcement
- Cross-functional alignment (Clinical, Regulatory, Data, Safety)
- Milestone integrity and protection of capital efficiency

05
04
03
02
01
CRA
Clinical
Trial
Manager
Trial
Manager
Project
Manager
Manager
Programme
Director
Director
Chief Operating Officer



Executive oversight with end-to-end functional visibility:
- Clinical Operations
- Regulatory & Compliance
- Data & Biostatistics
- Safety & Medical Oversight
- Quality & Vendor Governance
Including outsourced functions and vendor-delivered workstreams. Governance-led alignment, with direct stakeholder engagement when execution integrity requires it.
This dual perspective enables objective CRO performance assessment, early detection of execution drift, and structured outsourcing governance instead of reactive intervention. For biotech sponsors, this translates into controlled partnerships — not unmanaged delegation — and clear executive-level ownership across European delivery.
sponsor-side perspective
Enterprise risk exposure, governance accountability, and financial / investor consequence.
CRO-side leadership
Operational realities, capacity constraints, and vendor performance behavior.
Cross-Functional Governance Control
“I understand where functions must align, where accountability gaps form, and how execution drift begins. Governance is not reporting — it is structured accountability.
I can read programme reporting and see where accountability gaps emerge — sponsor-side or vendor-side.”
I can read programme reporting and see where accountability gaps emerge — sponsor-side or vendor-side.”
Strategic Leverage Across Sponsor and CRO Structures
executive depth & structural control
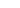
49+
countries managed
42+
visited personally
- Feasibility
- Start-up
- Enrollment & Treatment
- Database Lock
- CSR
Continuity across transition points protects programme integrity, capital efficiency, and regulatory positioning.
Phase I–IV leadership across global programmes, with strategic concentration in oncology.
Phase I–IV & End-to-End Programme Continuity
Strategic and programme-level leadership concentrated in Oncology, where complex design, global site networks and regulatory scrutiny require disciplined governance and risk control.
Oncology
delivery scope
- Rare disease
- Metabolic disease
- Inflammation disease (RA)
- Immunology
- Psychiatry
- Vaccines
- Neurology
- Dermatology
Experience across 17+ therapeutic areas, including:
Therapeutic Breadth with Oncology Depth






















Executive oversight experience across internal, GCP, vendor, sponsor, and financial audits.
Governance frameworks are built with inspection readiness in mind — reducing risk through structured accountability.
Governance frameworks are built with inspection readiness in mind — reducing risk through structured accountability.
regulatory & audit exposure
scalable regional operating extension
Structured access to additional regional capacity when programme scope expands.
- Activated only when required
- Operates under executive governance control
- Avoids permanent headcount expansion
- Maintains sponsor-side accountability
Best suited for: rapid geographic expansion or temporary workload peaks.
fractional governance advisory
Board- and investor-level advisory support focused on programme risk, outsourcing control, and governance alignment.
- Part-time executive engagement
- Independent programme risk assessment
- CRO performance evaluation
- Governance model review and optimisation
Best suited for: sponsors seeking structured oversight without operational replacement.
Interim Executive Programme Director
Full executive ownership
of a European-region clinical programme during stabilisation, expansion, or high-risk phases.
of a European-region clinical programme during stabilisation, expansion, or high-risk phases.
- Minimum engagement: typically, ≥0.6 FTE
- Direct governance authority across CROs and vendors
- Sponsor representation across the wider European region (including adjacent markets)
- Accountable for programme control, risk visibility, and delivery stability
Best suited for: programmes requiring immediate stabilisation, restructuring of oversight, or executive-level control without permanent hiring.
executive engagement model
Please complete the form and I'll get back to you shortly.
Interim / Contract European-region Programme Director
independent board & investor advisory
outsourced clinical talent
